Acetone Polar or Nonpolar Solvent
Acetone is a polar substance because of polarity in the carbonyl group due to the difference in the electronegativity of oxygen and carbon atom. In contrast in eg.
Polar Vs Nonpolar Solvents Identifications And Examples Psiberg
Ad Bulk Chemical Resin Supplier.

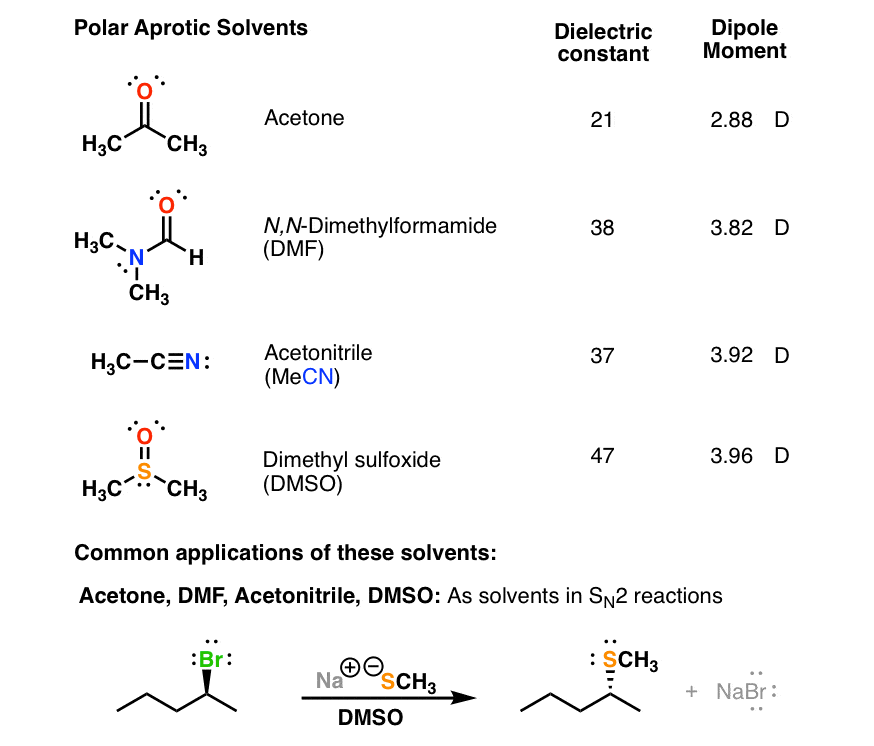
. The values in the table below except as noted have been extracted from online and hardbound compilations. Given acetones difference in polarity no wonder its such a great solvent. Acetone is a polar aprotic solvent.
Not all organic solvents are nonpolar. Polar solvent is a type of solvent that has large partial charges or dipole moments. Acetone has a dipole moment of 288 D and hence is a polar molecule despite possessing characteristics of both the polar and nonpolar substances.
The values for acetone are µ 288 D and ε 21. Acetone Dicholoroethane Tetrahydrofuran Dicholoromethane Chloroform Diethylether Benzene Toluene Xylene Carbontetrachloride Cyclohexane Petroleum ether Hexane Pentane Non-polar Polar. Is acetone a nonpolar solvent.
ScCO 2 is a promising green solvent because of its negligible toxicity and high solubility window for different kind of solutes. NMR Chemical Shifts of Common Laboratory Solvents as Trace Impurities. A solvent is polar if it has a dipole moment greater than 16 D and a dielectric constant greater than 5.
To unlock this. Christian Reichardt Solvents. See answer 1 Best Answer.
Is ethanol polar or nonpolar and why. Ad Products empowering scientists at every stage helping to deliver scientific breakthroughs. Contact Us for a Quote Now.
Acetones chemical makeup includes elements that are both polar and nonpolar which means acetone can be used with both organic and inorganic substances. Leading life science supplier for your research development or production needs. Chloroform is nonpolar because it has a low dielectric constant.
There are more and less polar solvents. So acetone is more polar than say hexane benzene and chloroform but less polar than acetonitrile acetic acid water and a saturated ammonium chloride solution. Polar solvents have a positive and a negative charge at different places in their structures and will dissolve other polar substances.
Acetones chemical makeup includes elements that are both polar and nonpolar which means acetone can be used with both organic and inorganic substances. So acetone is a polar solvent. Up to 24 cash back Is acetone polar or nonpolar solvent Acetone is an organic compound with its chemical formula CH32CO.
As you can see it has mixed with both the polar water molecules and. What is solvent polarity. It is classified as the simplest ketone.
Acetone is a relatively polar organic. Values for relative polarity eluant strength threshold limits and vapor pressure have been extracted from. Many students may have a question regarding whether acetone is polar or not.
Ad Recycle Acetone Save Money Save the Environment. Nonpolar solvents include alkanes pentane. So Is Acetone Polar or Nonpolar.
A polar solvent can dissolve ions and other polar compounds. It can use its H atom to participate in H-bonding. It has a characteristic odor and flammable in nature.
The bonds between the atoms have very different but measurable electronegativities. Any small molecule cannot be polar or non polar at the same time polarity is a range. Polar solvents can be used to dissolve inorganic or ionic compounds such as salts.
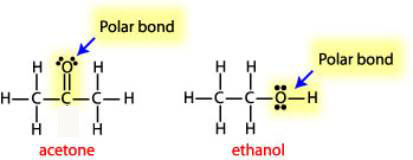
A protic solvent has an H atom bound to O or N. An example of a polar solvent is water while ethanol is more nonpolar. Is acetone or ethanol more polar.
Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances while other solvents can only dissolve one or the other. Acetone is a good solvent due to its ability to dissolve both polar and nonpolar substances while other solvents can only dissolve one or the other. Acetone is another molecular material with both polar and nonpolar characteristics.
Approved by eNotes Editorial Team. Why is chloroform a non-polar solvent. For example acetone C 3 H 6 O is an organic solvent but is polar as well.
It exists as a colorless volatile liquid. As a result the dipole moment of Acetone is around 269 D. The dielectric constant is the measure of the polarity of a solvent.
Acetone exists in a liquid state at room temperature. Acetone polar or nonpolar. Polarity of Solvents Author.
Water is a polar solvent. Why is acetone a better solvent than water. Acetone is a relatively polar organic solvent.
Other polar solvents include acetone acetonitrile dimethylformamide DMF dimelthylsulfoxide DMSO isopropanol and methanol. That is polar solvents like to mingle with polar solvents and the same applies to non-polar solvents. Here acetone has been added to water left tube and carbon tetrachloride right tube.
Water is a popular example of a polar solvent.

Why Is Acetone A Good Solvent Properties Explanation Video Lesson Transcript Study Com

Is C3h6o Acetone Polar Or Non Polar Youtube

Polar Protic Polar Aprotic Nonpolar All About Solvents
What Makes Acetone A Really Good Solvent What Allows It To Dissolve Both Polar And Non Polar Molecules Quora
Comments
Post a Comment